- BACTEROMIC
Our Solution
MEET BACTEROMIC - A UNIQUE DIAGNOSTIC SOLUTION
The comprehensive AST assay based on microfluidics design
Automated system with competitive unit costs and 60-panel throughput
Intended use: isolates and positive blood cultures*
* in development
Single panel for G+ G-
Determination of Real MIC for each antibiotic
Just two simple steps to prepare the sample
Integrated with existing systems
Easy-to-use
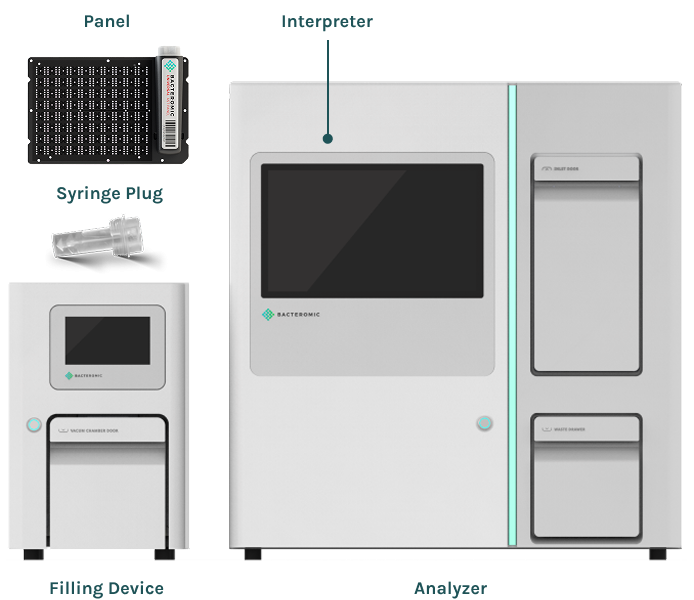
BACTEROMIC – Faster Access to Precise and Effective Diagnostics
The BACTEROMIC is an Antimicrobial Susceptibility Testing (AST) system that can identify the antibiotic resistances present in a certain bacterial infections, determine antibiotic efficacy against bacteria, and provide both susceptibility assessment and minimum inhibitory concentrations (MIC) of antibiotics in a single fast, automated procedure.
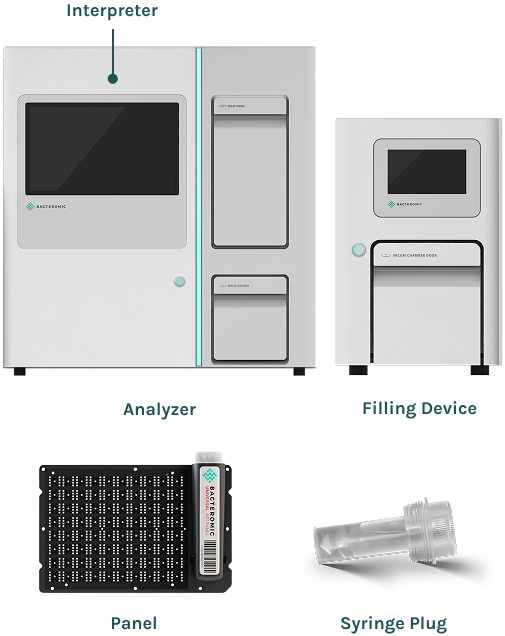
The BACTEROMIC system consists of 3 components (Filling Device, Analyzer, and Interpreter) and is used with BACTEROMIC Panels and accessory BACTEROMIC Syringe Plug. As an automated system, it simplifies and shortens laboratory procedures used in evaluating the drug susceptibility of microorganisms.
Microfluidic solutions are the heart of BACTEROMIC system.
The BACTEROMIC uses the most trusted, gold-standard microdilution antibiotic susceptibility testing method, combined with cutting-edge microfluidic solutions that allow for unprecedented information density on a single disposable panel.
BACTEROMIC Analyzer 2.0
BACTEROMIC Analyzer 2.0. is part of a BACTEROMIC System, which supports antimicrobial susceptibility testing of bacteria in an automated manner. Analyzer is responsible for providing necessary environmental conditions, data storage and presentation of results. It is intended for use only for medical laboratories personnel licensed to perform in vitro diagnostic tests. BACTEROMIC Interpreter ISW 2.0 as a part of the BACTEROMIC System is intended to be used as an aid for comprehensive assessment of bacterial susceptibility in people with bacterial infection.
BACTEROMIC Filling Device 2.0
BACTEROMIC Filling Device 2.0 is part of a BACTEROMIC system, which supports antimicrobial susceptibility testing of bacteria in an automated manner. The Filling Device’s function is to fill the Panel with a suspension of microorganisms and then seal it. It is intended for use only for medical laboratories personnel licensed to perform in vitro diagnostic tests.
BACTEROMIC Interpreter ISW 2.0
BACTEROMIC Interpreter ISW 2.0 is a fully automated software that quantitatively determines the value of the Minimum Inhibitory Concentration (MIC). Interpreter also qualitatively assesses the drug susceptibility of the tested organism assigning one of the three resistance categories to the test substance (S, I, R) and qualitatively assesses the presence or absence of a resistance mechanism involving the production of Extended-Spectrum Beta-Lactamases (ESBL) of the tested microorganism isolated from any biological material of human origin. It is intended for use only for medical laboratories personnel licensed to perform in vitro diagnostic tests.
BACTEROMIC Panel UNI 2.0
BACTEROMIC Panel UNI 2.0 is intended to be used with the BACTEROMIC System as an aid for comprehensive assessment of bacterial susceptibility in people with bacterial infection. BACTEROMIC Panel UNI 2.0 allows for the incubation of the microorganism isolated from any biological material of human origin, in the presence of a wide range of antimicrobials concentrations and then for the assessment of its growth. It is intended for use only for medical laboratories personnel licensed to perform in vitro diagnostic tests.
BACTEROMIC Syringe Plug
BACTEROMIC Syringe Plug is intended as an accessory to the BACTEROMIC Panel. Syringe Plug primary function is to seal the Panel, ensuring its proper operation according to its intended purpose. It is intended for use only for medical laboratories personnel licensed to perform in vitro diagnostic tests.
BACTEROMIC offers a comprehensive antibiogram.
Highest information capacity
640 incubation chambers
High
efficiency
up to 60 panels simultaneously in a single test
Fully compliant
with the
EUCAST guidelines
Assessment of the real (MIC)
across a wide range of concentrations – 6-14 per each antibiotic

Diagnostic panel
One panel – a complete set of reliable information
The BACTEROMIC system allows for a one-time identification of the effectiveness of a range of 30 antibiotics used in the treatment of, among others, respiratory, blood, urinary tract, and skin infections. It provides a quick and accurate assessment of the antibiotic susceptibility of both G+ G- bacteria in a single diagnostic panel.
BACTEROMIC Panel UNI 2.0 is intended to be used with the BACTEROMIC System as an aid for comprehensive assessment of bacterial susceptibility in people with bacterial infection. BACTEROMIC Panel UNI 2.0 allows for the incubation of the microorganism isolated from any biological material of human origin, in the presence of a wide range of antimicrobials concentrations and then for the assessment of its growth.
Single panel for G+ and G- bacteria

Each incubation chamber is connected to a chamber that serves as an air reservoir to ensure the growth of aerobic bacteria.

The incubation chambers are separated by a non-polar liquid, creating a barrier that prevents the diffusion of antibiotics.
BACTEROMIC Panel UNI 2.0
30 antibiotics + ESBL resistance mechanism
CE-IVDR
No of panels
One panel for G+ and G- bacteria (20 Gram +, 22 Gram -)
Time of AST
16 h
No of antibiotics
30 antibiotics + ESBL resistance mechanism
BACTEROMIC Panel UNI 2.0 - List of resistance mechanism tests
BACTEROMIC Panel UNI 2.0
List of antibiotics (G+ and G-) by group
Aminoglycosides
Diaminopyrimidines
Carbapenems
Lincosamides
Fluoroquinolones
Nitrofurans
Glycopeptides
Penicillinis
Cephalosporins
Macrolides
Oxazolidinones
BACTEROMIC Panel UNI FAST
Sample type
Bacteria Isolates
Time of AST
6 – 8 h (same shift results)
NEW PANELS ARE COMING SOON
BACTEROMIC Panel PBC
In development
For research use only – not for diagnostic use. These products are intended solely for presentation or demonstration purposes and cannot be made available until compliance with Regulation (EU) 2017/746 is ensured.
Sample type
Positive Blood Culture
Time of AST
6 – 8 h (same shift results)
Procedure for use
The BACTEROMIC workflow is fully compatible with the traditional workflow of hospital laboratories. For isolates as a starting sample it consists of six steps, four of them are done manually and two are fully automatic.

0,5 McFarland bacteria suspension is prepared from single colony.

Bacteria solution is 300x diluted in MH broth.

Bacterial solution is pipetted onto the panel.

Panels are automatically filled & sealed for the analysis.

Panels are transferred from Filling Device into Analyzer.

Analyzer performs automated AST on up to 60 cartridges.

Technology

Clinical Advisory Board

Target Audience
BACTEROMIC is an excellent solution for:
Hospitals
Central
Labs
Microbiology
Labs
Unique combination of technologies used in the BACTEROMIC system provides for a variety of potential applications
Universal
Panel
Drug susceptibility assessment directly from positive blood culture
Assessment of synergistic effects of antibiotics
Customized AST Panel
Assessment of drug susceptibility of animal pathogens
Development
2026
- BACTEROMIC System with UNI Panel obtained CE IVDR Certification
2025
- Publication of Analytical Performance Evaluation of BACTEROMIC system results
2024
- Scope Fluidics launches Analytical Performance Evaluation of BACTEROMIC system at second center
- BACTEROMIC system 2.0 under IVDR certification process
2023
- BACTEROMIC secures U.S. Patent for Ultra-Fast Antimicrobial Susceptibility Testing Method and System
- Scope Fluidics launches Analytical Performance Evaluation for BACTEROMIC system
2022
- Development of BACTEROMIC system 2.0
- BACTEROMIC Panel UNI composition update
- Implementation of the Analytical Performance Evaluation
2021
- Obtaining the ISO 13485:2016 certification
- Clinical validation of the BACTEROMIC system
Early Access Program
performance data
Precision
In the BACTEROMIC precisely determined the minimum inhibitory concentration (MIC) in tested range of concentrations for 96% of antibiotic–bacteria pairs, compared to 38% in competing solutions.
Bacteromic data on file, 2026.
Reliability
EA and CA agreement rates against very common automated AST being 93% and 94%, respectively, for both GN and GP bacteria underscore the high quality and consistency of our panels.
Bacteromic data on file, 2026.
Next Steps
With this significant milestone achieved, we’re moving forward with further verifications in Europe and outside, bringing us closer to delivering an even more advanced diagnostic tool worldwide.



The project has received funding from the European Union's HORIZON 2020 research and innovation programme under contract no. 881101.

Obtaining international patent protection for the inventions ‘Microfluidic chip and Incubation segment’



